Dr. Nogah Nativ
Department of Communication Disorders

Research
Dr. Nogah Nativ
Department of Communication Disorders

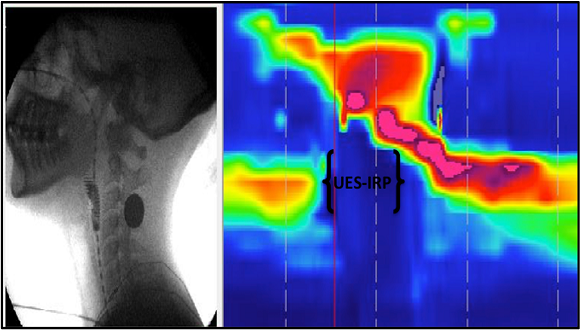
Swallowing disorders are prevalent in older adults and can result in significant morbidity and mortality. Consequences of dysphagia (difficulty swallowing) include malnutrition, dehydration, social isolation, pneumonia, and reduced quality of life.
Dr. Nativ Zeltzer’s research focuses on the effects of ageing on the swallow and devising prophylactic treatment methods for the preservation of swallow function in the elderly. The lab utilizes high resolution manometry and videofluoroscopic biomechanical analysis of the swallow to characterize physiological components of both normal and disordered swallowing, with the goal of designing accurately targeted preventative and rehabilitative treatment for adults with dysphagia. An additional area of focus is the identification of risk factors and biomarkers for the development of aspiration pneumonia, a common complication of swallowing disorders, facilitating the design of sensitive diagnostic tools for early detection of aspiration pneumonia.

Research
Dr. Yuval Nir, Ph.D.
Department of Physiology and Pharmacology

Sleep is a universal behavior that is present across the animal kingdom. We spend a third of our lives sleeping, disconnected from the world around us. Our sleep is closely regulated so that when we are sleep deprived, we ultimately compensate with longer, deeper sleep. Sleep helps our cognitive performance, promoting learning and memory consolidation. Lack of sleep immediately affects our cognition, mood, and health. All this suggests that sleep is essential, but what exactly is it about brain activity during sleep that is so crucial for restoring our normal cognition?
Sleep also involves dramatic changes to our perceptual awareness. Sometimes our consciousness fades altogether while at other times we experience vivid dreams. Although our brain continues to be active, we are mostly disconnected from sensory signals such as sounds, which would otherwise be perceived, trigger plasticity and result in behavior. How does the internal state of brain activity during sleep affect brain responsiveness and perceptual awareness?
Our goal is to understand how sleep relates to cognition and perception. Our research is guided by a belief that such studies require a combination of human and animal models. We therefore use multiple experimental techniques, focusing on the strengths of each setup to investigate the same key questions synergistically. Animal models are used to investigate underlying mechanisms, by performing detailed recordings of electrical activity and by manipulating neuronal activity with optogenetic, electrical and sensory stimulation. Human studies are carried out for careful investigation of cognitive factors and for studying large-scale brain activity (with fMRI, EEG, recordings in neurosurgical patients, and behavioral tests).
Research
Prof. Ilana Lotan, Ph.D.
Department of Physiology and Pharmacology

Main research projects currently in the lab:
Research methods:
Research
Prof. Dario G. Liebermann, Ph.D.
Department of Physical Therapy

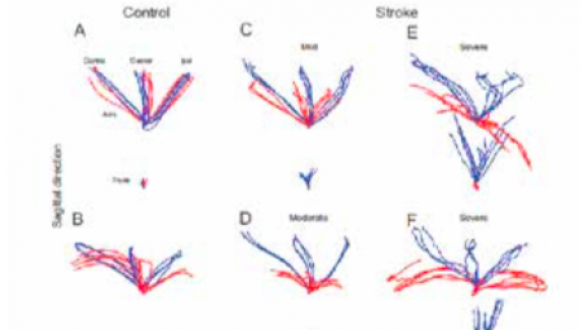
Behavioral and computational motor control is our field of research. This is a main venue for understanding the motor system and its organization, in healthy and clinical populations.
In the last years, we have dedicated major efforts in investigating methods and technologies (virtual reality, robot-based rehabilitation, neuro-stimulation) that can potentially enhance motor recovery and functional performance in clinical populations with a focus on upper-limb motion in stroke survivors.
Mathematical model-based, as well as empirical neuromotor approaches, are used in our research for studying and understanding laws of motor control and sensorimotor integration.
Research
Prof. Talma Hendler, M.D., Ph.D.
Department of Physiology and Pharmacology

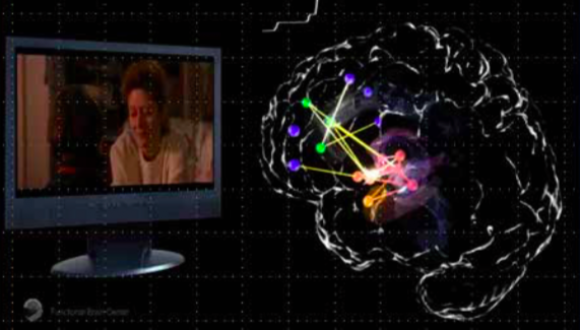
Investigating brain mechanisms underlie generation and regulation of the human emotional experience, in healthy and pathological states. The research is based on measuring indices of brain structure and functional dynamics via MRI (functional-MRI, DTI and Volumetric-MRI) and separate or simultaneous recording of electrical signals (scalp-EEG and intracranial-EEG).
The characterization of individual brain response is based on correlating neural activity and connectivity with behavioral and physiological measurements of emotionality (e.g. heart rate, hormone secretion, genetic expression, skin conductance, eye movements and verbal output). Induction of emotional states is achieved via film and music media, inter-personal interactions, and interactive social games. Regulation of emotions is modulated via on-line feedback protocols from brain signals in a closed loop set-up (i.e. NeuroFeedback).
The lab is also involved in studies aim to advance translation while focusing on neural markers of vulnerability and recovery with regard to post traumatic disorders (e.g. anxiety and depression), developmental disorders (e.g. schizophrenia and personality) and neurodegenerative disorders (e.g. Parkinson's Disease).
An essential part of this aspect of our work is the development of advanced new tools for acquiring and analyzing whole brain neural measurements; including applying multi-scale mapping for capturing dynamics of brain networks.
Research
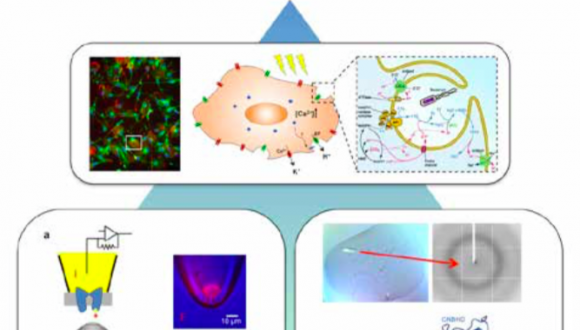
Dr. Yoni Haitin, Ph.D.
Department of Physiology and Pharmacology

Projects in the lab include:
Research
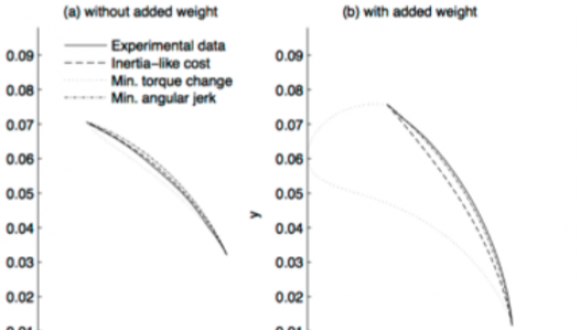
Dr. Jason Friedman, Ph.D.
Department of Physical Therapy

We study human movement in typical and clinical populations, with a focus on grasping and finger movements.
We are interested in fundamental questions such as how we learn to make new movements, how children develop motor skills during development, and how our motor function is affected by disorders such as stroke, dystonia or cerebral palsy. We also study the interconnection between decision making and human movements.
Our approach is to construct models that describe movement and force generation by the hand and arm, taking into account the biomechanics of the hand and the neural processes leading up to making movements. This approach gives us insights into the strategies behind the complex movements and force coordination required to successfully perform grasping and manipulation, as well as a greater understanding of the causes of differences in performance in individuals with motor disorders.
A goal of this research is to improve rehabilitation of hand function through improving our knowledge of these strategies.
Research
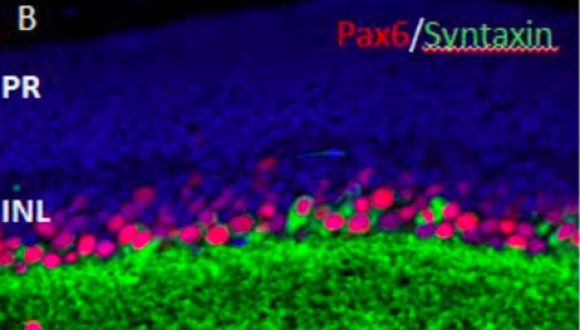
Prof. Ruth Ashery-Padan, Ph.D.
Department of Human Molecular Genetics and Biochemistry

We study the gene networks that transform the embryonic cells into a complex, differentiated organ. We focus on exploring this question by studying the process of eye development as a model for organogenesis.
We apply cutting-edge technologies including mouse genetic tools (Cre/loxP), molecular biology, and microarray analysis to identify and functionally characterize genes that regulate the development of the eye in mammals.
Understanding the normal developmental regulation of the different eye structures is essential for understanding visual disorders and designing treatments for ocular phenotypes including retinal degeneration, glaucoma and cataracts, all of which are leading causes of blindness.